The Galvanizing Process

Galv With Us is a blogpost where we at Metals & Chemicals Technology Sdn Bhd share our insights on the galvanizing industry. We often share such insights with our customers in the form of newsletters and now we would like to share with others as well
THE GALVANIZING PROCESS

Galvanization has been a fundamental rust prevention process since the early 1700. The process of galvanizing is the application of coating iron or steel metals with molten zinc to resist corrosion from taking place. Initially, such applications have been recorded to take place as early as 10 B.C. when civilization were applying zinc to copper to create brass alloy. However, only in 1742 a French chemist known as P. J. Melouin (Figure 1) produced a study to explain the achievement of zinc coating when iron is dipped in molten zinc which led to the discovery of hot-dip galvanizing.

The galvanizing process (Figure 4) begins from pre-treatment stages or also known as ‘Surface Preparation’ where steel has to be properly cleaned to ensure a smooth and quality bonding between the molten zinc and the metal surface takes place when dipped into the kettle.
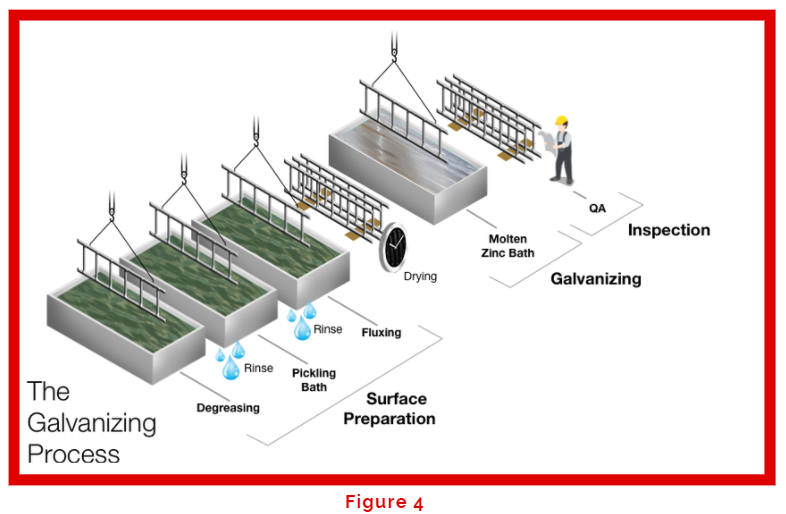
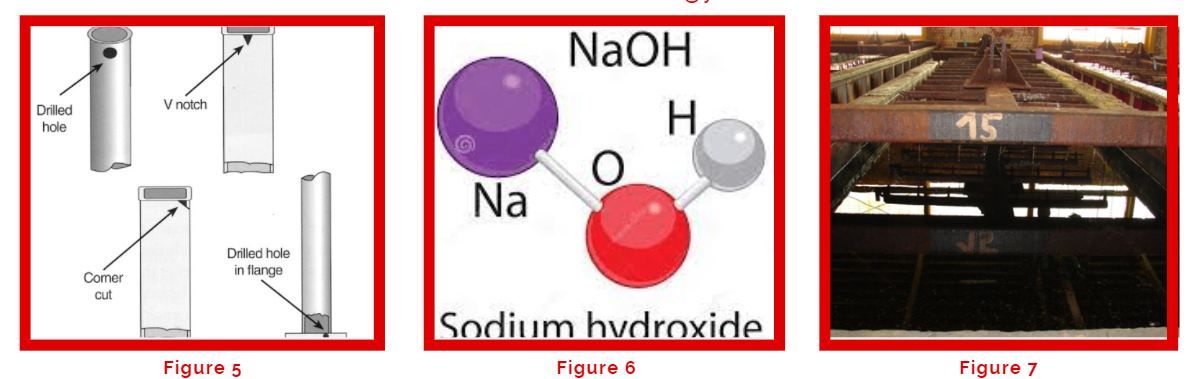
If you look at any galvanizing process, the QA inspection is listed as the final step. However, the first step in the pre-treatment process is the QA checking of the structures that are to be cleaned and galvanized. Every galvanizer should ensure the structures are properly vented for drainage (Figure 5) to avoid any chemicals or zinc to be accumulated within the structures that could cause re-galvanization. Other important parameters to ensure are structure’s size and shape fit into the kettle, welding areas and slags have been cleaned, and proper marking to distinguish the different structures or materials. After that is the degreasing step that removes oil and grease which is commonly achieved with the use of caustic soda (NaOH) (Figure 6) at 10% concentration and temperature of 60°C. Another degreasing method would be the use of an acid degreaser but isn’t as effective as an alkaline degreaser (Figure 7) in removing oil and grease. If proper QA inspection (Figure 8) isn’t done in the beginning, then degreasing will not be 100% effective which leads to poor surface preparation in the following steps hence overall leading to a possible re-galvanization which is costly. Once the structures go through degreasing, they are then rinsed properly with water before being dipped into the pickling bath.
Articles are further cleaned in the pickling process, the 15% hydrochloric acid (HCl) removes scales, carbons and rust providing a contaminant free surface. With proper degreasing and rinsing, galvanizers are able to save cost with less bottlenecking, lower acid consumption and optimized conditions. Pickling baths are able to operate at temperatures of 20-40°C with additives such as AV Ultra (supplied by Metals & Chemicals Technology) which also reduces fuming, reduces floating oil (Figure 9), acts as a degreaser, inhibitor to reduce over-pickling and prevents rusting before fluxing. The fourth step in the pre-treatment process is again the rinsing step. Rinsing is essential in the pre-treatment as it avoids carry over of unwanted chemicals and contaminants into the next stage.


Then, the structures go into a flux solution which operates at 50-70°C. The flux solution removes any oxide film which forms on the steel surface. A proper fluxing creates a lower surface tension on the steel thus providing an easy drag out of solutions (Figure 10). Next, the articles are taken out and dried completely to avoid any defect formation during galvanizing in the zinc bath. The zinc bath (Figure 11) operates at temperatures of 450-460°C. Therefore, it is essential that the articles are properly cleaned and dried out otherwise the zinc bath will experiencing flames (Figure 12). Once the structures are galvanized, they then go through a passivation process to prevent the formation of zinc oxides.


